A HoloLens application for pre-operative patient planning was approved by the American Food and Drug Administration FDA.
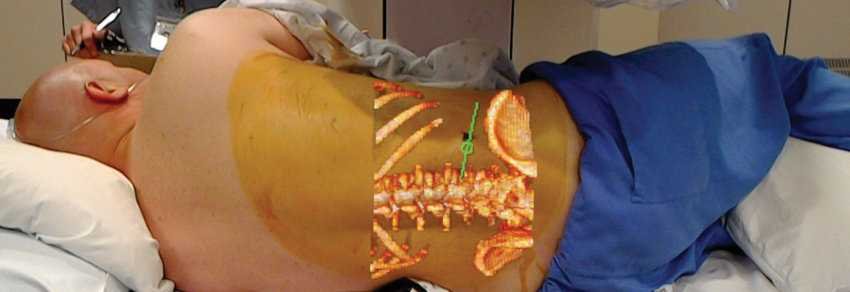
The HoloLens application OpenSight Augmented Reality System is one augmented reality for surgical applications, manufactured by Novarad, and enables surgeons to create interactive 2D, 3D and even 4D images of their patients' anatomy by projecting them accurately onto the patient's body to allow accurate surgical planning.
Unlike previous pre-operative planning systems they use virtual actually, OpenSight uses Microsoft HoloLens® glasses, which allow doctors to view 3D images of patients in augmented reality, highlight relevant anatomy and avoid critical structures, while also placing virtual tools and guidance systems to more accurately plan a surgical approach.
Overlapping holograms can be viewed simultaneously by multiples users of the HoloLens glasses, which makes them useful in both clinical and educational settings. In addition, an educational version of it is provided software available for medical students to perform mock dissections on cadavers.
See a relevant video below: